Kelvin

The Unit – kelvin, K (kelvin)
The SI unit of temperature, the kelvin (K), is named after the Belfast-born engineer and physicist William Thomson, who became Lord Kelvin in 1892 as a recognition of his achievements in thermodynamics.
For more than 60 years, the kelvin was defined as the fraction 1/273.16 of the thermodynamic temperature (TTPW) of the triple point of water – a unique temperature at which the three phases of water (solid, liquid, and vapour) co-exist in equilibrium. As part of a wider redefinition of the SI that came into effect in May 2019, the kelvin’s definition was changed, and is now expressed in terms of a fixed value of the Boltzmann constant, k. The official definition is as follows:
“The kelvin, symbol K, is the SI unit of thermodynamic temperature. It is defined by taking the fixed numerical value of the Boltzmann constant k to be 1.380 649 × 10–23 when expressed in the unit J K–1, which is equal to kg m2 s–2 K–1, where the kilogram, metre and second are defined in terms of h, c and ΔνCs.”
The Boltzmann constant turns up in all physical formulae that describe the physical effects caused by temperature. This results from the way temperature is described – as a thermodynamic concept that characterises the average kinetic energy of small particles in thermal equilibrium. One such relationship is Boyle’s law:
PV = nRT ,
which relates the pressure and volume of n atoms of an ideal gas to its temperature. Unfortunately, neither gas thermometers, which exploit Boyle’s Law, nor most other thermodynamic thermometers, are sufficiently small or convenient to be of practical use, and these thermometers have only recently become accurate enough to satisfy the needs of science and industry. The best gas thermometer experiments take several months of work, and a million dollars-worth of equipment, to measure a single temperature.
To overcome this limitation, the General Conference on Weights and Measures (CGPM) defined a more practical scale that is based on defined temperatures for the melting, freezing, and triple points of pure substances. In addition, they approved interpolating thermometers to define the temperatures that fall between these points. This scale, which is reproducible to about 1 mK for temperatures below a few hundred degrees Celsius, is updated approximately every 20 years. The current version is called the International Temperature Scale of 1990, or ITS-90(external link). Temperatures on ITS-90 can be expressed either in kelvin or in degrees Celsius; these are sometimes distinguished using an upper-case T and lower-case t, respectively. The two units have the same size (so that temperature differences or intervals are equal in both units), but values are offset from each other:
T / K = t/°C +273.15
Differences between the ITS-90 temperature scale and thermodynamic temperature can be found here(external link).
The definition of the kelvin that came into being on World Metrology Day 2019 will allow for the future development of technologies that may improve the accuracy of temperature measurement, free of any limitations arising from the use of triple point of water cells. This is particularly important for measurements well away from TTPW – i.e., at very low (below ~20 K) and very high (above ~1300 K) temperatures – as the uncertainties increase with extrapolation away from the triple point of water. At these extreme temperatures, the practicality of thermodynamic measurement has already been demonstrated and these methods can now take advantage of the new definition to achieve uncertainties competitive with ITS-90. However, for most of the temperature range, the triple point of water is likely to remain the fundamental practical reference for the foreseeable future, and ITS-90 is expected to remain in force for many decades to come.
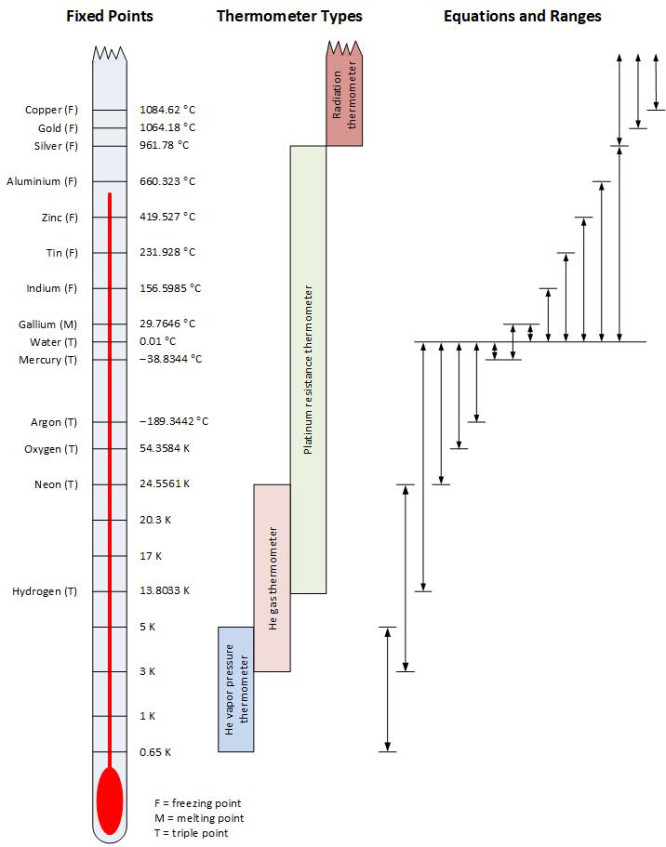
ITS-90 temperature scale showing the fixed points, the interpolating thermometer types, and ranges over which different interpolating equations are used. MSL covers ranges from the argon point to the copper point.
Technical Capability
We are leading experts in contact thermometry, radiation thermometry, and humidity, and provide a wide range of temperature-related measurement and consultancy services. We can help you to understand limitations in the accuracy of industrial thermometers, identify errors in applications of radiation thermometry, or improve the accuracy of humidity measurements.
We provide a calibration service covering the temperature ranges –190 °C to 550 °C for contact thermometry and –25 °C to 1100 °C for radiation thermometry, and the relative humidity range 10 % RH to 95 % RH from –60 °C to 70 °C (see here for our Temperature and Humidity calibration services)
Our Research
Our research covers a wide range of topics, from difficult industrial measurements, sensor characterisation, new calibration techniques and instruments, physical modelling and uncertainty analysis, to the fundamental definitions of the standards. Research topics include:
- Reflection errors in radiation thermometry, for applications in the petrochemical, metal processing, and food industries. This includes the effects of emissivity and the bidirectional reflectance distribution function (BRDF) of industrial materials.
- Limitations in the accuracy of industrial thermometers – immersion effects, hysteresis, and ageing effects in thermocouples.
- Using thermocouple homogeneity scanning to develop an understanding of the damage mechanisms in thermocouples, and to develop new stable rare metal reference thermocouples.
- Calibration equations and propagation of uncertainty for standard platinum resistance thermometers (SPRTs), radiation thermometers, and humidity sensors.
- Propagation of uncertainty and covariance, using generalised least squares, in the IAPWS-95 thermodynamic equations of state for pure water and associated properties.
- Development of a Resistance Bridge Calibrator (RBC) based on a combinatorial technique using four stable base resistors.
- Size-of-source effect (SSE) and non-linearity in radiation thermometers.
- Effects of isotopic composition and glass dissolution on the triple point of water.
- New definition of relative humidity based on fugacity.
- Use of double-wavelength radiation thermometry to measure thermodynamic temperature.
- Measurement of Boltzmann’s constant using Johnson noise thermometry.
Relevant Publications
Jifeng Qu, Samuel P Benz, Kevin Coakley, Horst Rogalla, Weston L Tew, Rod White, Kunli Zhou, Zhenyu Zhou, “An improved electronic determination of the Boltzmann constant by Johnson noise thermometry”, Metrologia, 54, 549–558, 2017.
B Fellmuth, J Fischer, G Machin, SPicard, P P M Steur, O Tamura, D R White, H Yoon, “The kelvin redefinition and its Mise en Pratique”, Philosophical Transactions of the Royal Society A. Mathematical, Physical and Engineering Sciences, 374, article no. 20150037, 2016.
R Feistel, R Wielgosz, S A Bell, M F Camões, J R Cooper, P Dexter, A G Dickson, P Fisicaro, A H Harvey, M Heinonen, O Hellmuth, H-J Kretzschmar, J W Lovell-Smith, T J McDougall, R Pawlowicz, P Ridout, S Seitz, P Spitzer, D Stoica, H Wolf, “Metrological challenges for measurements of key climatological observables: oceanic salinity and pH, and atmospheric humidity. Part 1: overview”, Metrologia, 53, R1–R11, 2016.
J W Lovell-Smith, R Feistel, A H Harvey, O Hellmuth, S A Bell, M Heinonen, J R Cooper, “Metrological challenges for measurements of key climatological observables. Part 4: atmospheric relative humidity”, Metrologia, 53, R40–R59, 2016.
D R White, “Some mathematical properties of the ITS-90”, in Temperature: Its Measurement and Control in Science and Industry, Vol. 8, edited by C W Meyer, AIP Conference Proceedings, Melville, New York, 81–88, 2013.
P Saunders, “Uncertainties in the realisation of thermodynamic temperature above the silver point”, International Journal of Thermophysics, 32, 26–44, 2011.
G Machin, P Bloembergen, K Anhalt, J Hartmann, M Sadli, P Saunders, E Woolliams, Y Yamada, H Yoon, "Practical implementation of the Mise en Pratique for the definition of the kelvin above the silver point", International Journal of Thermophysics, 31, 1779–1788, 2010.
D R White, W L Tew, “Improved estimates of the isotopic correction constants for the triple point of water”, International Journal of Thermophysics, 31, 1644–1653, 2010.
D R White, M Ballico, D del Campo, S Duris, E Filipe, “Uncertainties in the realization of the SPRT sub-ranges of the ITS-90”, International Journal of Thermophysics, 28, 1868–1881, 2007.
Go to our Temperature calibration & services page.
View a short video about the Kelvin here(external link).

